![pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom](https://i.pinimg.com/736x/99/80/05/998005d7b3fbb74f7a91222f3209e7c5--physical-chemistry-ap-chemistry.jpg)
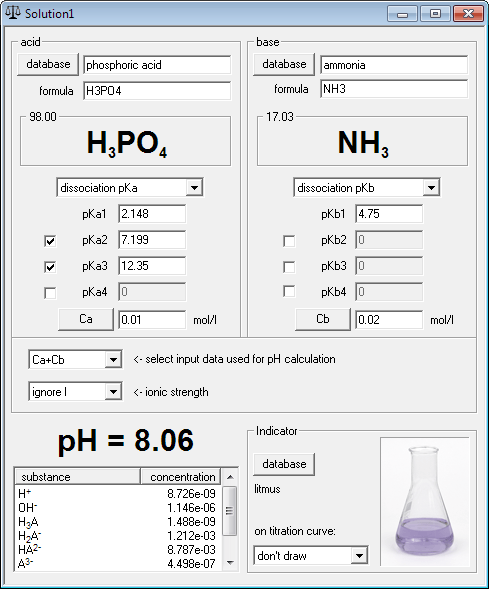
pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom

pH calculator for both strong and weak (Monoprotic) acids | Online pH calculator for both strong and weak (Monoprotic) acids App/Software Converter – CalcTown




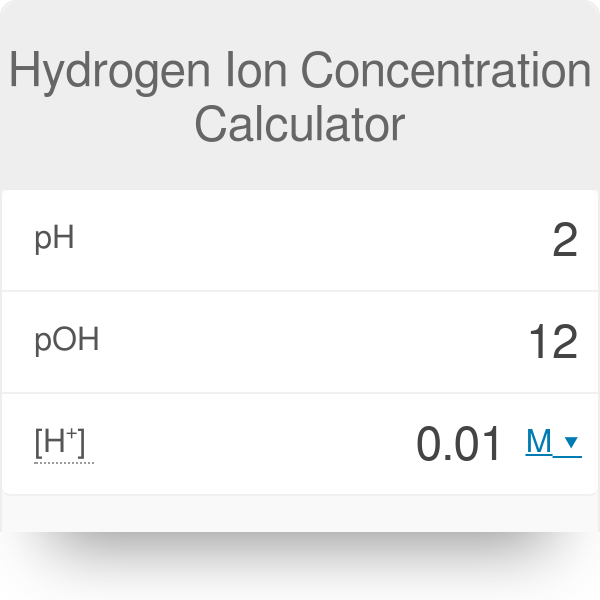












![Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC" Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC"](http://itsactuallyquitebasic.weebly.com/uploads/2/7/8/0/27808159/4950515.png?357)