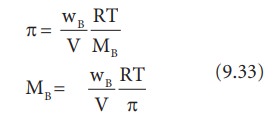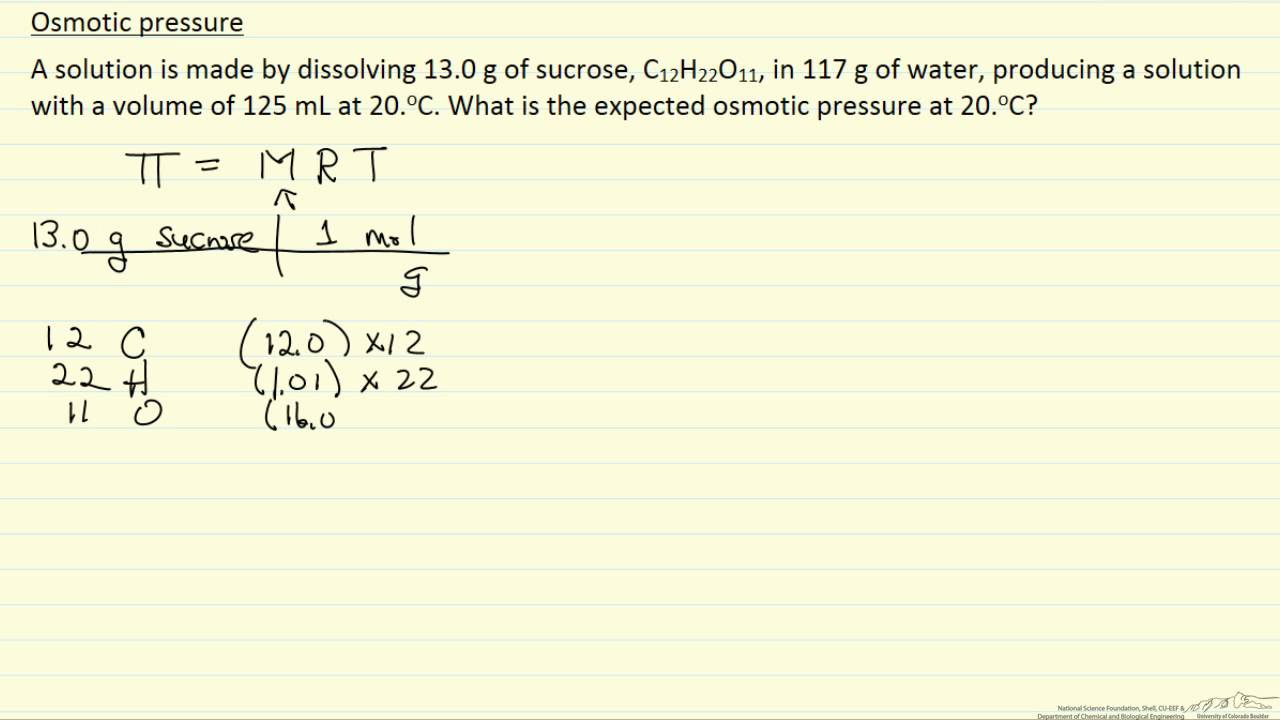Calculate the osmotic pressure of 5% solution of urea at 272 K (R = 0.0821 L-atm. deg^-1) - Sarthaks eConnect | Largest Online Education Community
How can the equation for osmotic pressure be basically the same as the ideal gas equation? Aren't the molecules in a liquid moving much slower? - Quora

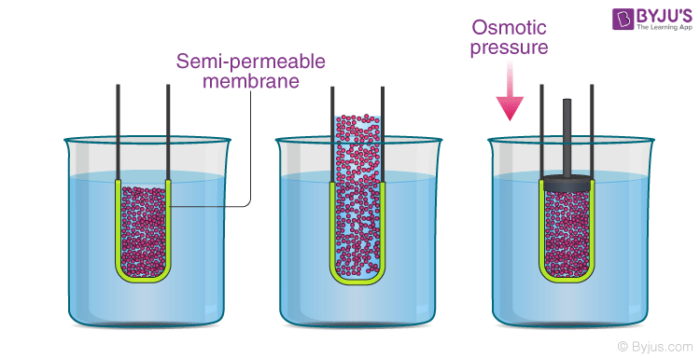
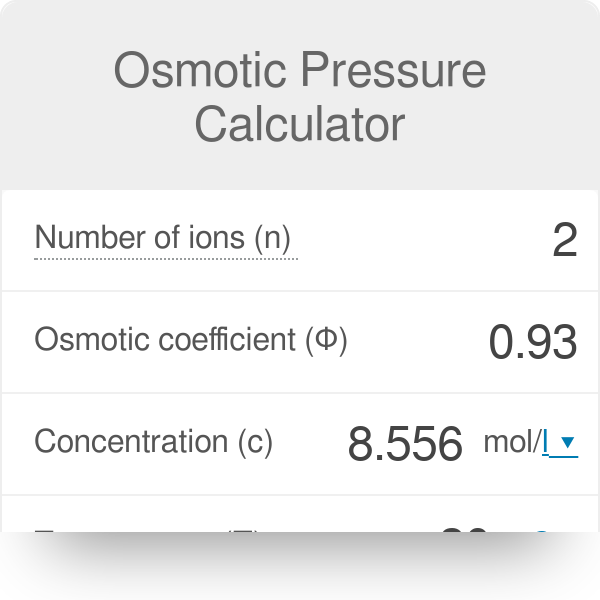
Hi need help! in calculating the osmotic pressure using the formula, does the solute conc. mean in the feed or permeate? : r/WaterTreatment

Calculate the osmotic pressure of a solution obtained by mixing 100 cm^(3) of 1.5% solution of urea (mol. Mass=60) and 100 cm^(3) of 3.42% solution by cane sugar (mol. Mass = 342)

The osmotic pressure of a solution (density is 1 g `mL^(-1))` containing `3 g` of glucose (molec... - YouTube

PPT - Drill: Calculate the osmotic pressure of 5.0 g NaOH in 7500 mL soln at 27 o C. PowerPoint Presentation - ID:4784680
Calculate the osmotic pressure of 5% solution of urea at 273K. - Sarthaks eConnect | Largest Online Education Community
![Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ] Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ]](https://i.ytimg.com/vi/PtV0tDX6lCI/maxresdefault.jpg)
Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ]
![For a 5% solution of urea (Molar mass - 60 g/mol), calculate the osmotic pressure at 300 K. [R = 0.0821 L atm K^(-1) mol^(-1)] For a 5% solution of urea (Molar mass - 60 g/mol), calculate the osmotic pressure at 300 K. [R = 0.0821 L atm K^(-1) mol^(-1)]](https://d10lpgp6xz60nq.cloudfront.net/ss/web/1522124.jpg)